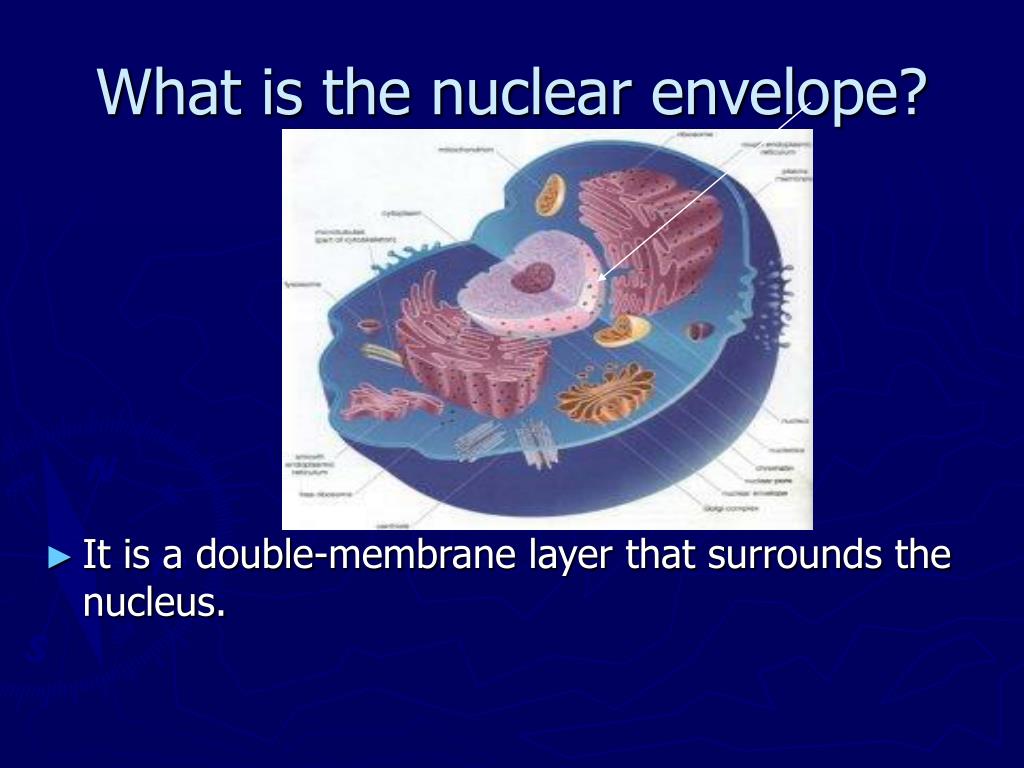
In several cellular processes mediated by ESCRT, ESCRT-III proteins are recruited both by the ESCRT-I/II complexes and/or the Bro1-domain containing protein Alix ( Henne et al., 2011 Schöneberg et al., 2017). The polymers are then remodeled by the ATPases Associated with diverse cellular Activities (AAA ATPase) Vps4 to promote membrane scission. Using process-specific cues, the ESCRT-III proteins are recruited to different sites of action and assemble into polymers. 
ESCRT is best known for mediating scission of membranes that bud away from the cytoplasm ( Christ et al., 2017 McCullough et al., 2018 Schöneberg et al., 2017 Scourfield and Martin-Serrano, 2017). Recent studies have suggested that the endosomal sorting complex required for transport (ESCRT) machinery plays a major role in many of the above described aspects of NE dynamics ( Frost et al., 2012 Radulovic et al., 2018). Understanding the mechanisms underlying NE dynamics thus has broad implications for cell and organismal physiology. Failure to establish or maintain NE integrity compromises genome integrity, as occurs during aging, cellular senescence, cancer, or cell migration through confined spaces ( Bakhoum et al., 2018 Spektor et al., 2017 Umbreit and Pellman, 2017 Ungricht and Kutay, 2017). Finally, pathological “rupture” of the NE, which can then undergo membrane repair, occurs in a variety of circumstances ( Halfmann et al., 2019 Hatch and Hetzer, 2014 Shah et al., 2017). NE remodeling also occurs during interphase nuclear pore complex insertion in all eukaryotes ( Grossman et al., 2012 Ungricht and Kutay, 2017) and for spindle pole body (SPB) insertion and/or removal in yeast ( Cavanaugh and Jaspersen, 2017 Ding et al., 1997). In metazoans, the NE is disassembled, partially or completely, at the beginning of mitosis and reassembled after the completion of chromosome segregation. During cell division, the NE undergoes complex dynamics, necessitating mechanisms for controlled remodeling. In eukaryotes, the nuclear envelope (NE) separates the nucleoplasm and cytoplasm. These findings establish the existence of mechanisms for NE sealing independent of the canonical ESCRT pathway. Additional results suggest a potentially general role for unsaturated fatty acids in NE integrity. A gain-of-function mutation implicated a membrane and ESCRT-III regulator, Alx1, in this alternate pathway. In the absence of Cmp7, Lem2 formed aggregates that appear to interfere with ESCRT-independent NE sealing. Unexpectedly, loss of Lem2 or its interacting partner Nur1 suppressed cmp7Δ defects. A genetic screen identified mutations that promote NE integrity in cmp7Δ cells. japonicus cells lacking Cmp7 have compromised NE sealing after mitosis but are viable. To identify such mechanisms, we studied NE assembly in Schizosaccharomyces japonicus, a fission yeast that undergoes partial mitotic NE breakdown and reassembly. However, prior work has hinted at CHMP7/ESCRT-independent mechanisms. Subsequently, the chromosomes enter the daughter cell where the spindle is formed, and the remnant of the NE collapses into the mother cell.The nuclear envelope (NE) undergoes dynamic remodeling to maintain NE integrity, a process involving the inner nuclear membrane protein LEM2 recruiting CHMP7/Cmp7 and then ESCRT-III. There is an early increase in permeability, suggestive of pore remodeling, and then an obvious opening in the NE near the spindle pole body (yellow). ( d) In Ustilago maydis, a basidiomycete, the NE is dragged from the mother cell to the bud by microtubules and dynein (black). Dynein-mediated movement is thought to then pull the NE toward the centrosomes, eventually causing a rupture at a distal region of the NE. ( c) In human tissue culture cells (somatic), microtubules originating from the centrosomes (yellow) connect to the NE via the microtubule motor dynein (black). Local concentration of this coatomer complex may then lead to vesiculation of the NE, as depicted, or to a non-conventional role for COPI. ( b) In embryonic-like nuclei formed in vitro from Xenopus egg extract, nuclear pore proteins recruit the COPI complex (red) to the NE. During the second phase of disassembly, larger holes in the NE are proposed to emanate from the site of disassembled pores. ( a) In starfish oocytes, early alterations in permeability at the nuclear pore (green) have been observed, and correlate with an early phase of nuclear pore complex (NPC) disassembly. Cells are not drawn to scale it is notable that the oocyte is very large compared to a somatic cell, since size may impose specific constraints on the mechanics of NE breakdown. Key findings in various experimental systems are schematically depicted. Mechanistic models of nuclear envelope (NE) disassembly.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
